
The cytoskeleton (likewise CSK) is a cellular “ scaffolding” or “ skeletal system” included within a cell’s cytoplasm and also is constructed of healthy protein.
The cytoskeleton exists in all cells; it was when believed to be special to eukaryotes, yet the current research study has actually recognized the prokaryotic cytoskeleton. What does the cytoskeleton do in the plasma membrane?

It has frameworks such as flagella, cilia and also lamellipodia and also plays essential duties in both intracellular transportation (the activity of blisters and also organelles, for instance) and also the mobile department.
In 1903 Nikolai K Koltsov recommended that the form of cells was identified by a network of tubules that he called the cytoskeleton.
The principle of a healthy protein mosaic that dynamically collaborated cytoplasmic biochemistry and biology was recommended by Rudolph Peters in 1929 while the term (cytosquelette, in French) was first presented by French embryologist Paul Wintrebert in 1931.
Eukaryotic Cytoskeleton
Eukaryotic cells include 3 primary sorts of cytoskeletal filaments, which are microfilaments, intermediate filaments, and also microtubules.
The cytoskeleton supplies the cell with the framework and also form, and also by leaving out macromolecules from several of the cytosols it includes in the degree of macromolecular extensively in this area.
Cytoskeletal components communicate thoroughly and also thoroughly with mobile membrane layers.
A variety of little particle cytoskeletal medications has actually been found that communicate with actin and also microtubules.
These substances have actually verified helpful in researching the cytoskeleton and also numerous have professional applications.
In animal cells, the cytoskeleton maintains the three-dimensional structure and helps the cell.
1. Microfilaments (actin filaments)
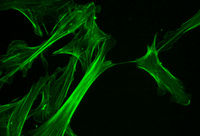
These are the thickest filaments of the cytoskeleton. They are made up of straight polymers of actin subunits and also create pressure by prolongation at one end of the filament combined with shrinking at the various other, creating internet activity of the interfering hair.
They likewise serve as tracks for the activity of myosin particles that affix to the microfilament and also “stroll” among them. Actin structures are regulated by the Rho household of little GTP-binding healthy proteins such as Rho itself for contractile actomyosin filaments (” stress and anxiety fibers”), Rac for lamellipodia and also Cdc42 for filopodia. The cytoskeleton consists of microtubules, microfilaments, and intermediate filaments.
2. Intermediate filaments
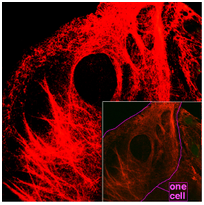
These filaments, balancing 10 nanometers in size, are extra steady (highly bound) compared to actin filaments, and also heterogeneous components of the cytoskeleton. Like actin filaments, they work in the upkeep of cell-shape by birthing stress (microtubules, by comparison, withstand compression.
It might be useful to consider mini- and also intermediate filaments as cable televisions, and also of microtubules as mobile support beam of lights). Intermediate filaments arrange the inner tridimensional framework of the cell, securing organelles and also functioning as architectural elements of the nuclear lamina and also sarcomeres. They likewise take part in some cell-cell and also cell-matrix joints.
Various intermediate filaments
- made from vimentins, being the typical architectural assistance of several cells.
- made from keratin, discovered in skin cells, hair and also nails.
- Neurofilaments of neural cells.
- Made from lamin, providing architectural assistance to the nuclear envelope.
3. Microtubules

Microtubules are hollow cylindrical tubes regarding 23 nm in size (lumen = about 15 nm in size), a lot of typically making up 13 protofilaments which, then, are polymers of alpha and also beta-tubulin. They have a really vibrant practice, binding GTP for polymerization. They are typically arranged by the centrosome.
In 9 triplet collections (star-shaped), they create the centrioles, and also in 9 doublets oriented regarding 2 added microtubules (wheel-shaped) they create cilia and also flagella.
The last development is typically described as a “9 +2” plan, wherein each doublet is attached to one more by the healthy protein dynein.
- DNA Damage: Causes, Consequences, and Repair Mechanisms
- Gene Silencing: Cellular Mechanisms and Applications
- Euchromatin: The Dynamic Realm of Gene Expression
- Heterochromatin: From Chromosome to Protein
As both flagella and also cilia are architectural elements of the cell, and also are kept by microtubules, they could be thought about a component of the cytoskeleton.
They play essential duties in:
- Intracellular transportation (related to dyneins and also kinesins, they move organelles like mitochondria or blisters).
- the axoneme of cilia and also flagella.
- the mitotic pin.
- synthesis of the cell wall surface in plants.
Prokaryotic Cytoskeleton
The cytoskeleton was formerly believed to be a function just of eukaryotic cells, yet homologs to all the significant healthy proteins of the eukaryotic cytoskeleton have actually lately been discovered in prokaryotes.
Although the transformative connections are so far-off that they are not noticeable from healthy protein series contrasts alone, the resemblance of their three-dimensional frameworks and also comparable features in keeping cell form and also polarity supplies solid proof that the eukaryotic and also prokaryotic cytoskeletons are absolutely homologous.
Nevertheless, some frameworks in the microbial cytoskeleton might have yet to be recognized.
1. FtsZ
FtsZ was the initial healthy protein of the prokaryotic cytoskeleton to be recognized. Like tubulin, FtsZ types filaments in the visibility of GTP, yet these filaments do not team right into tubules.
Throughout cellular division, FtsZ is the initial healthy protein to transfer to the department website, and also is important for hiring various other healthy proteins that manufacture the brand-new cell wall surface in between the splitting cells.
2. MreB and also ParM
Prokaryotic actin-like healthy proteins, such as MreB, are associated with the upkeep of cell form. All non-spherical germs have genetics inscribing actin-like healthy proteins, and also these healthy proteins create a helical network below the cell membrane layer that overviews the healthy proteins associated with cell wall surface biosynthesis.
Some plasmids inscribe a dividing system that includes an actin-like healthy protein ParM. Filaments of ParM display vibrant instability, and also might dividing plasmid DNA right into the splitting little girl cells by a system similar to that made use of by microtubules throughout eukaryotic mitosis.
3. Crescentin
The germs Caulobacter crescentus consists of a Third healthy protein, crescentin, that relates to the intermediate filaments of eukaryotic cells.
Crescentin is likewise associated with keeping cell form, such as helical and also vibrioid types of germs, yet the system through which it does this is presently vague.
