
Passive transport occurs when no energy is required to move a substance, such as water or carbon dioxide, from an area of high concentration to an area of low concentration until the concentration is equal, sometimes across a membrane. The high-to-low concentration gradient is the driving force for passive transport because it fulfils a fundamental law of nature: Things tend to move from a high-energy, ordered structure to a lower-energy, increasing randomness, or increasing entropy state of being.

What is passive transport?
Passive transport definition
Passive transport is a movement of ions and other atomic or molecular substances across cell membranes without the need of energy input. Unlike active transport, it does not require an input of cellular energy because it is instead driven by the tendency of the system to grow in entropy.
Types of Passive Transport
The following types of passive transport system are available at the cellular membrane level.
1. Diffusion
This is a good example of how certain molecules, such as oxygen, simply move directly through a membrane in response to the high-to-low concentration gradient.
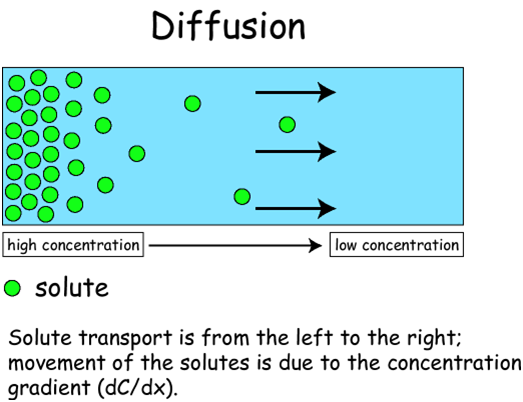
As an example, oxygen diffuses out of the lungs and into the blood for transport to all of the cells.
2. Facilitated Diffusion
This is a special type of diffusion that is useful because substances are sometimes too large to move freely through a membrane, or they need to move against a concentration gradient so transport proteins embedded in the membrane assist with the passage.
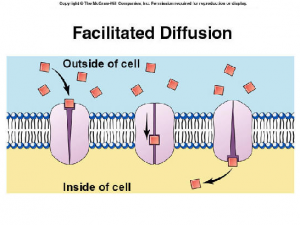
In most cases, the transport protein creates a chemical channel for the passage of a specific substance. Because no energy is expended, the rate of facilitated diffusion depends on the number of transport proteins embedded in the membrane.
As an example, glucose is moved by a glucose-transporter protein as it passes through the red blood cell into a body cell.
3. Osmosis
This is similar to diffusion except that it refers only to water diffusing through a permeable membrane. Water as a solvent moves from an area of high to low concentration.
In biological systems, it is easier to think of water as flowing from a low-solute to a high solute concentration until the concentration is equal. The solution that has a high solute concentration is a hypotonic solution relative to another lower solute concentration or hypertonic solution.
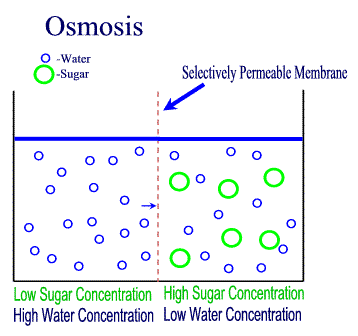
Water will continue to osmotically move from the low-solute/high solvent concentration toward the high-solute/low solvent concentration until both sides are isotonic, or equal. Osmoregulation is a struggle for all organisms as we continually adjust our cellular water balance for optimal conditions.
In your body, the large intestine reabsorbs water by osmosis to help maintain the proper water concentration, which helps to keep your systems from dehydrating.
4. Ion channels
These are membrane proteins that allow the passage of ions that would ordinarily be stopped by the lipid bilayer of the membrane. These small passageways are specific for one type of ion, such that a calcium ion could not pass through an iron ion channel.
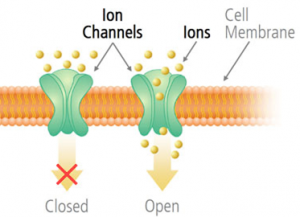
The ion channels also serve as gates because they regulate ion flow in response to two environmental factors: chemical or electrical signals from the cells and membrane movement. This happens in your body when a nervous impulse encounters a gap or synaptic cleft between nerve cells.
The electrical stimulation is continued because ion channels are opened to allow specific ions to pass through the receiving membrane, which continues the electrical stimulation to the next nerve cell.
What is the difference between active and passive transport?
- Active transport pumps molecules through the cell membrane against the concentration gradient
- Utilizes cellular energy in the form of ATP
- Endocytosis, exocytosis, secretion of substances into the bloodstream and sodium/potassium pump are types of active transport.
- Allows molecules to pass the cell membrane, disrupting the equilibrium established by the diffusion.
- Ions, large proteins, complex sugars, and cells are transported.
- Required for the entrance of large, insoluble molecules into the cell.
Passive Transport
- Passive transport allows molecules to pass the cell membrane through a concentration gradient
- Does not require cellular energy
- Diffusion, facilitated diffusion and osmosis are the types of passive transport
- A dynamic equilibrium of water, nutrients, gases, and waste is maintained between cytosol and the extracellular environment
- Water-soluble molecules like small monosaccharides, lipids, carbon dioxide, oxygen, and water are transported
- Allows the maintenance of delicate homeostasis between the cytosol and extracellular fluid
Additional Readings:
